Abstract
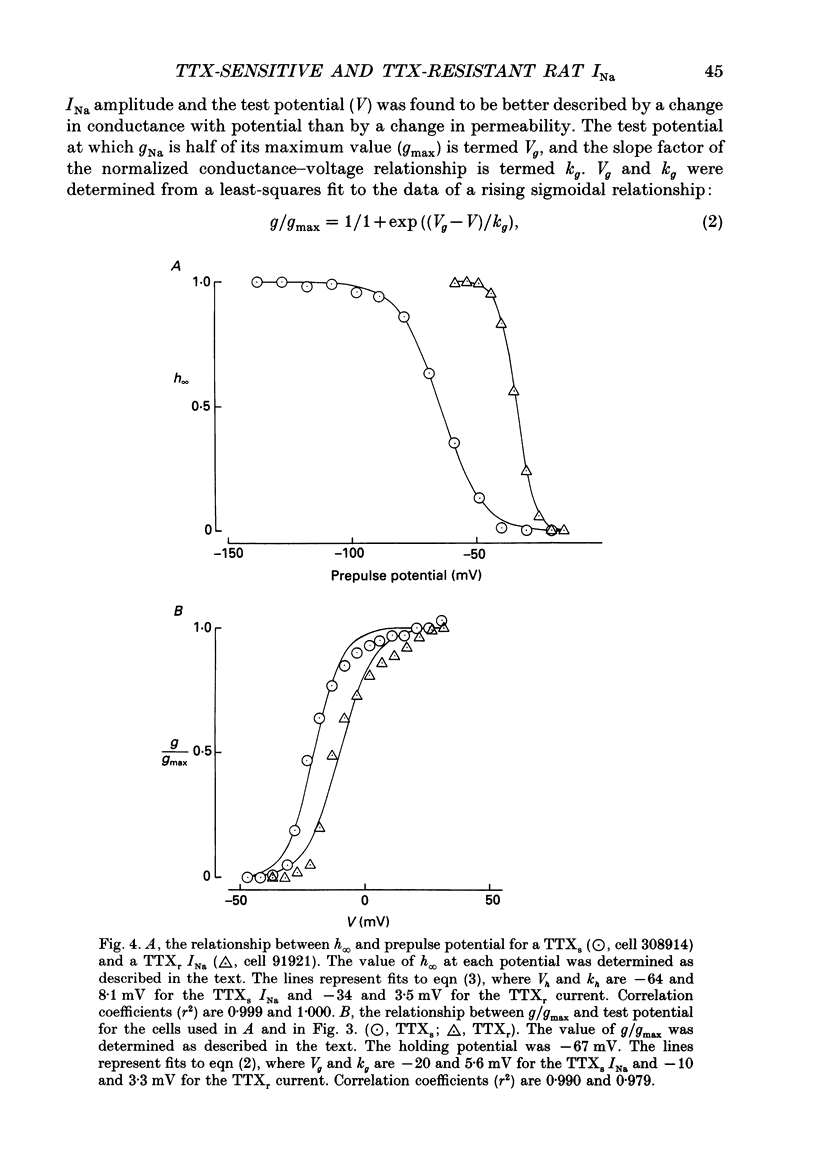
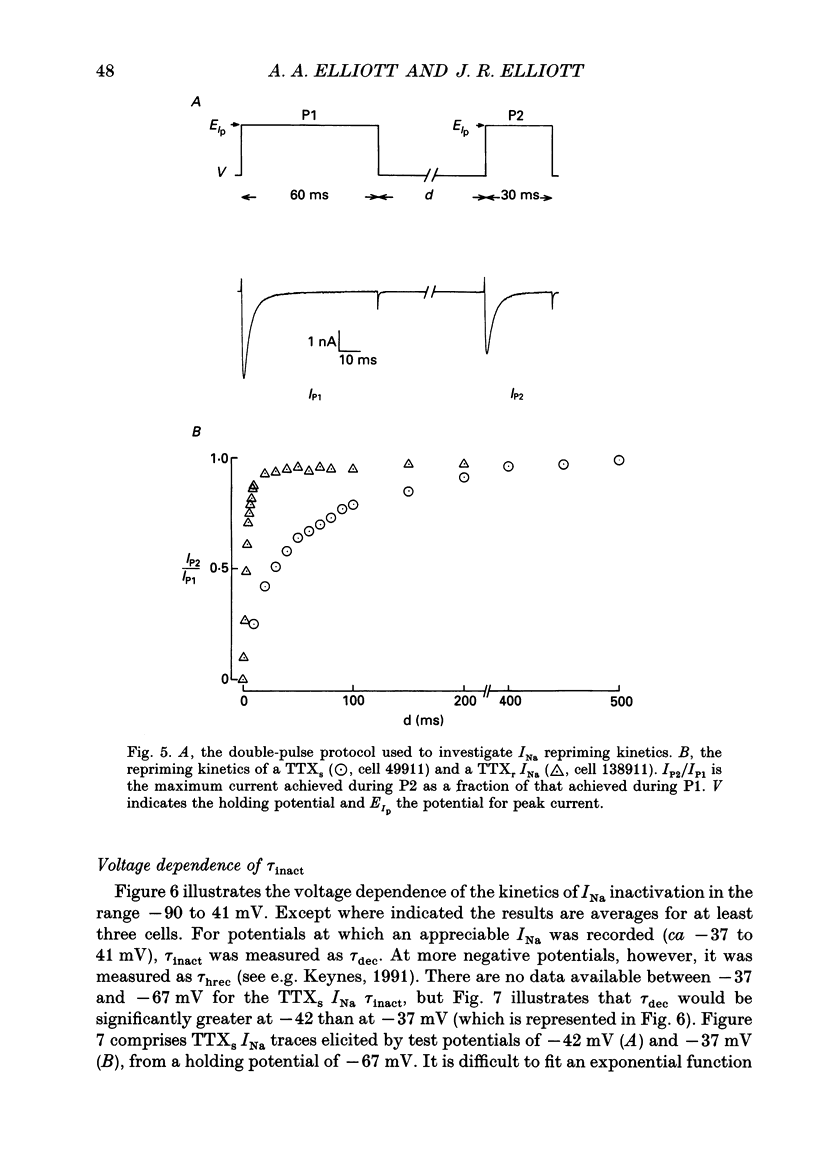
1. The whole-cell patch-clamp technique was used to investigate the characteristics of two types of sodium current (INa) recorded at room temperature from small diameter (13-25 microns) dorsal root ganglion (DRG) cells, isolated from adult rats and maintained overnight in culture. 2. Sodium currents were isolated pharmacologically. Internal Cs+ and external tetraethylammonium (TEA) ions were used to suppress potassium currents. A combination of internal EGTA, internal F-, a low (10 microM) concentration of external Ca2+ and a relatively high (5 mM) concentration of internal and external Mg2+ was used to block calcium channels. The remaining voltage-dependent currents reversed direction at the calculated sodium equilibrium potential. Both the reversal potential and magnitude of the currents exhibited the expected dependence on the external sodium concentration. 3. INa subtypes were characterized initially in terms of their sensitivity to tetrodotoxin (TTX). TTX-sensitive (TTXs) currents were at least 97% suppressed by 0.1 microM TTX. TTX-resistant (TTXr) INa were recorded in the presence of 0.3 microM TTX and appeared to be reduced in amplitude by less than 50% in 75 microM TTX (n = 1). 4. As in earlier studies, the peak of the current-voltage relationship, the mid-point of the normalized conductance curve and the potential (Vh) at which the steady-state inactivation parameter (h infinity) was 0.5 were found to be significantly more depolarized for the TTXr INa (by ca 10, 14 and 37 mV respectively). There was little difference in the slope at the mid-point of the normalized conductance curves (the mean slope factors were 5.1 mV for the TTXs INa and 4.9 mV for the TTXr current) but the h infinity curves for TTXr currents were significantly steeper than those for TTXs currents (mean slope factors of 3.8 and 11.5 mV respectively). Both the time to peak and the decay time constant of the peak current recorded from a holding potential of -67 mV were more than a factor of three slower for the TTXr INa than for the TTXs current. 5. However, in direct contrast to the difference in activation and decay kinetics, 'slow' TTXr INa recovered from inactivation at -67mV, or reprimed, more than a factor of ten faster than 'fast' TTXs INa. 6. The differences apparent in both the repriming kinetics of TTXs and TTXr INa at -67 mV and the kinetics of the decay phase of the peak INa are shown to be explicable largely in terms of the voltage dependence of their respective inactivation systems.(ABSTRACT TRUNCATED AT 400 WORDS)
Full text
PDF















Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baccaglini P. I., Cooper E. Electrophysiological studies of new-born rat nodose neurones in cell culture. J Physiol. 1982 Mar;324:429–439. doi: 10.1113/jphysiol.1982.sp014122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benoit E., Corbier A., Dubois J. M. Evidence for two transient sodium currents in the frog node of Ranvier. J Physiol. 1985 Apr;361:339–360. doi: 10.1113/jphysiol.1985.sp015649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bossu J. L., Feltz A. Patch-clamp study of the tetrodotoxin-resistant sodium current in group C sensory neurones. Neurosci Lett. 1984 Oct 12;51(2):241–246. doi: 10.1016/0304-3940(84)90558-5. [DOI] [PubMed] [Google Scholar]
- Carbone E., Lux H. D. Omega-conotoxin blockade distinguishes Ca from Na permeable states in neuronal calcium channels. Pflugers Arch. 1988 Nov;413(1):14–22. doi: 10.1007/BF00581223. [DOI] [PubMed] [Google Scholar]
- Clark R. B., Tse A., Giles W. R. Electrophysiology of parasympathetic neurones isolated from the interatrial septum of bull-frog heart. J Physiol. 1990 Aug;427:89–125. doi: 10.1113/jphysiol.1990.sp018163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elliott A. A., Elliott J. R. The influence of charge on the effects of n-octyl derivatives on sodium current inactivation in rat sensory neurones. J Physiol. 1991;440:35–53. doi: 10.1113/jphysiol.1991.sp018694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fedulova S. A., Kostyuk P. G., Veselovsky N. S. Ionic mechanisms of electrical excitability in rat sensory neurons during postnatal ontogenesis. Neuroscience. 1991;41(1):303–309. doi: 10.1016/0306-4522(91)90219-e. [DOI] [PubMed] [Google Scholar]
- Fedulova S. A., Kostyuk P. G., Veselovsky N. S. Two types of calcium channels in the somatic membrane of new-born rat dorsal root ganglion neurones. J Physiol. 1985 Feb;359:431–446. doi: 10.1113/jphysiol.1985.sp015594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukuda J., Kameyama M. Tetrodotoxin-sensitive and tetrodotoxin-resistant sodium channels in tissue-cultured spinal ganglion neurons from adult mammals. Brain Res. 1980 Jan 20;182(1):191–197. doi: 10.1016/0006-8993(80)90844-6. [DOI] [PubMed] [Google Scholar]
- Gallego R. The ionic basis of action potentials in petrosal ganglion cells of the cat. J Physiol. 1983 Sep;342:591–602. doi: 10.1113/jphysiol.1983.sp014870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HODGKIN A. L., HUXLEY A. F. The dual effect of membrane potential on sodium conductance in the giant axon of Loligo. J Physiol. 1952 Apr;116(4):497–506. doi: 10.1113/jphysiol.1952.sp004719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamill O. P., Marty A., Neher E., Sakmann B., Sigworth F. J. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Arch. 1981 Aug;391(2):85–100. doi: 10.1007/BF00656997. [DOI] [PubMed] [Google Scholar]
- Harper A. A. Similarities between some properties of the soma and sensory receptors of primary afferent neurones. Exp Physiol. 1991 May;76(3):369–377. doi: 10.1113/expphysiol.1991.sp003504. [DOI] [PubMed] [Google Scholar]
- Hess P., Lansman J. B., Tsien R. W. Calcium channel selectivity for divalent and monovalent cations. Voltage and concentration dependence of single channel current in ventricular heart cells. J Gen Physiol. 1986 Sep;88(3):293–319. doi: 10.1085/jgp.88.3.293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heyer E. J., Macdonald R. L. Calcium- and sodium-dependent action potentials of mouse spinal cord and dorsal root ganglion neurons in cell culture. J Neurophysiol. 1982 Apr;47(4):641–655. doi: 10.1152/jn.1982.47.4.641. [DOI] [PubMed] [Google Scholar]
- Ikeda S. R., Schofield G. G. Tetrodotoxin-resistant sodium current of rat nodose neurones: monovalent cation selectivity and divalent cation block. J Physiol. 1987 Aug;389:255–270. doi: 10.1113/jphysiol.1987.sp016656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikeda S. R., Schofield G. G., Weight F. F. Na+ and Ca2+ currents of acutely isolated adult rat nodose ganglion cells. J Neurophysiol. 1986 Mar;55(3):527–539. doi: 10.1152/jn.1986.55.3.527. [DOI] [PubMed] [Google Scholar]
- Jones S. W. Sodium currents in dissociated bull-frog sympathetic neurones. J Physiol. 1987 Aug;389:605–627. doi: 10.1113/jphysiol.1987.sp016674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keynes R. D. On the voltage dependence of inactivation in the sodium channel of the squid giant axon. Proc Biol Sci. 1991 Jan 22;243(1306):47–53. doi: 10.1098/rspb.1991.0008. [DOI] [PubMed] [Google Scholar]
- Kostyuk P. G., Krishtal O. A. Effects of calcium and calcium-chelating agents on the inward and outward current in the membrane of mollusc neurones. J Physiol. 1977 Sep;270(3):569–580. doi: 10.1113/jphysiol.1977.sp011969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kostyuk P. G., Veselovsky N. S., Fedulova S. A. Ionic currents in the somatic membrane of rat dorsal root ganglion neurons-II. Calcium currents. Neuroscience. 1981;6(12):2431–2437. doi: 10.1016/0306-4522(81)90089-0. [DOI] [PubMed] [Google Scholar]
- Kostyuk P. G., Veselovsky N. S., Tsyndrenko A. Y. Ionic currents in the somatic membrane of rat dorsal root ganglion neurons-I. Sodium currents. Neuroscience. 1981;6(12):2423–2430. doi: 10.1016/0306-4522(81)90088-9. [DOI] [PubMed] [Google Scholar]
- Lee K. S., Akaike N., Brown A. M. Trypsin inhibits the action of tetrodotoxin on neurones. Nature. 1977 Feb 24;265(5596):751–753. doi: 10.1038/265751a0. [DOI] [PubMed] [Google Scholar]
- MeLean M. J., Bennett P. B., Thomas R. M. Subtypes of dorsal root ganglion neurons based on different inward currents as measured by whole-cell voltage clamp. Mol Cell Biochem. 1988 Mar-Apr;80(1-2):95–107. doi: 10.1007/BF00231008. [DOI] [PubMed] [Google Scholar]
- Meiri H., Spira G., Sammar M., Namir M., Schwartz A., Komoriya A., Kosower E. M., Palti Y. Mapping a region associated with Na channel inactivation using antibodies to a synthetic peptide corresponding to a part of the channel. Proc Natl Acad Sci U S A. 1987 Jul;84(14):5058–5062. doi: 10.1073/pnas.84.14.5058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noda M., Suzuki H., Numa S., Stühmer W. A single point mutation confers tetrodotoxin and saxitoxin insensitivity on the sodium channel II. FEBS Lett. 1989 Dec 18;259(1):213–216. doi: 10.1016/0014-5793(89)81531-5. [DOI] [PubMed] [Google Scholar]
- Omri G., Meiri H. Characterization of sodium currents in mammalian sensory neurons cultured in serum-free defined medium with and without nerve growth factor. J Membr Biol. 1990 Apr;115(1):13–29. doi: 10.1007/BF01869102. [DOI] [PubMed] [Google Scholar]
- Petersen M., Pierau F. K., Weyrich M. The influence of capsaicin on membrane currents in dorsal root ganglion neurones of guinea-pig and chicken. Pflugers Arch. 1987 Aug;409(4-5):403–410. doi: 10.1007/BF00583794. [DOI] [PubMed] [Google Scholar]
- Pröbstle T., Rüdel R., Ruppersberg J. P. Hodgkin-Huxley parameters of the sodium channels in human myoballs. Pflugers Arch. 1988 Aug;412(3):264–269. doi: 10.1007/BF00582507. [DOI] [PubMed] [Google Scholar]
- Roy M. L., Narahashi T. Differential properties of tetrodotoxin-sensitive and tetrodotoxin-resistant sodium channels in rat dorsal root ganglion neurons. J Neurosci. 1992 Jun;12(6):2104–2111. doi: 10.1523/JNEUROSCI.12-06-02104.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruppersberg J. P., Schure A., Rüdel R. Inactivation of TTX-sensitive and TTX-insensitive sodium channels of rat myoballs. Neurosci Lett. 1987 Jul 22;78(2):166–170. doi: 10.1016/0304-3940(87)90627-6. [DOI] [PubMed] [Google Scholar]
- Sah P., Gibb A. J., Gage P. W. The sodium current underlying action potentials in guinea pig hippocampal CA1 neurons. J Gen Physiol. 1988 Mar;91(3):373–398. doi: 10.1085/jgp.91.3.373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlichter R., Bader C. R., Bernheim L. Development of anomalous rectification (Ih) and of a tetrodotoxin-resistant sodium current in embryonic quail neurones. J Physiol. 1991 Oct;442:127–145. doi: 10.1113/jphysiol.1991.sp018786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schofield G. G., Ikeda S. R. Sodium and calcium currents of acutely isolated adult rat superior cervical ganglion neurons. Pflugers Arch. 1988 May;411(5):481–490. doi: 10.1007/BF00582368. [DOI] [PubMed] [Google Scholar]
- Schwartz A., Palti Y., Meiri H. Structural and developmental differences between three types of Na channels in dorsal root ganglion cells of newborn rats. J Membr Biol. 1990 Jun;116(2):117–128. doi: 10.1007/BF01868670. [DOI] [PubMed] [Google Scholar]
- Scroggs R. S., Fox A. P. Calcium current variation between acutely isolated adult rat dorsal root ganglion neurons of different size. J Physiol. 1992 Jan;445:639–658. doi: 10.1113/jphysiol.1992.sp018944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stansfeld C. E., Wallis D. I. Properties of visceral primary afferent neurons in the nodose ganglion of the rabbit. J Neurophysiol. 1985 Aug;54(2):245–260. doi: 10.1152/jn.1985.54.2.245. [DOI] [PubMed] [Google Scholar]
- Weiss R. E., Sidell N. Sodium currents during differentiation in a human neuroblastoma cell line. J Gen Physiol. 1991 Mar;97(3):521–539. doi: 10.1085/jgp.97.3.521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshida S., Matsuda Y., Samejima A. Tetrodotoxin-resistant sodium and calcium components of action potentials in dorsal root ganglion cells of the adult mouse. J Neurophysiol. 1978 Sep;41(5):1096–1106. doi: 10.1152/jn.1978.41.5.1096. [DOI] [PubMed] [Google Scholar]
- Yoshida S., Matsuda Y. Studies on sensory neurons of the mouse with intracellular-recording and horseradish peroxidase-injection techniques. J Neurophysiol. 1979 Jul;42(4):1134–1145. doi: 10.1152/jn.1979.42.4.1134. [DOI] [PubMed] [Google Scholar]


